
Sodium phenoxide when heated with CO$_2$, under pressure at 125$^\circ$C yields a product which on acetylation produces C.
The major product C would be:

(a)

(b)

(c)
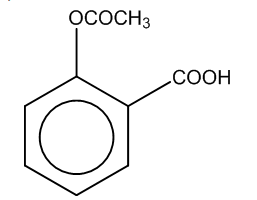
(d)





Answer
168.6k+ views
Hint: This question is based on the concept of acetylation reaction. As we can see in the question that B to C conversion takes place through the acetyl group. This is the Kolbe-Schmitt reaction, so now the product can be identified.
Complete step by step answer:
Now, we will move step by step for this reaction.
As we know, sodium phenoxide is the sodium salt of phenol.
The first step is when sodium phenoxide reacts with carbon-dioxide at high temp, and 5 atm to produce the salicylate i.e.

In the first step, we can see that phenol group is attached to the benzene ring, and COO- group is attached with the sodium ion.
Thus, the product formed in the first step is B.
Now, if we perform the further reaction to attain the product C, then there is a reaction of salicylate formed with the acid i.e. sulphuric acid in the presence of an acetyl group.
Thus, the product formed is

Here, we can see that –COOH group is attached at the ortho position, and the product formed is C i.e. salicylic acid.It is also known as aspirin.
The second step of this reaction is an example of acetylation reaction.
As mentioned the whole reaction i.e. from sodium phenoxide to the formation of salicylic acid is Kolbe-Schmitt reaction.
Hence, the major product formed C is

The correct option is (C).
Note: Don’t get confused while writing the complete chemical reaction, just perform it step by step as shown. There is a difference between salicylate, and salicylic acid. Salicylic acid is formed by the reaction of salicylate with sulphuric acid. So, salicylate acts as a starting material for acetylation reaction.
Complete step by step answer:
Now, we will move step by step for this reaction.
As we know, sodium phenoxide is the sodium salt of phenol.
The first step is when sodium phenoxide reacts with carbon-dioxide at high temp, and 5 atm to produce the salicylate i.e.

In the first step, we can see that phenol group is attached to the benzene ring, and COO- group is attached with the sodium ion.
Thus, the product formed in the first step is B.
Now, if we perform the further reaction to attain the product C, then there is a reaction of salicylate formed with the acid i.e. sulphuric acid in the presence of an acetyl group.
Thus, the product formed is

Here, we can see that –COOH group is attached at the ortho position, and the product formed is C i.e. salicylic acid.It is also known as aspirin.
The second step of this reaction is an example of acetylation reaction.
As mentioned the whole reaction i.e. from sodium phenoxide to the formation of salicylic acid is Kolbe-Schmitt reaction.
Hence, the major product formed C is

The correct option is (C).
Note: Don’t get confused while writing the complete chemical reaction, just perform it step by step as shown. There is a difference between salicylate, and salicylic acid. Salicylic acid is formed by the reaction of salicylate with sulphuric acid. So, salicylate acts as a starting material for acetylation reaction.
Recently Updated Pages
Hydrocarbons: Types, Formula, Structure & Examples Explained

Classification of Elements and Periodicity in Properties | Trends, Notes & FAQs

JEE Main 2021 July 25 Shift 1 Question Paper with Answer Key

JEE Main 2021 July 22 Shift 2 Question Paper with Answer Key

Sandmeyer Reaction Mechanism: Steps, Diagram & Exam Notes

Environmental Chemistry Chapter for JEE Main Chemistry

Trending doubts
JEE Main 2025 Session 2: Application Form (Out), Exam Dates (Released), Eligibility, & More

Displacement-Time Graph and Velocity-Time Graph for JEE

Atomic Structure - Electrons, Protons, Neutrons and Atomic Models

JEE Main 2025: Derivation of Equation of Trajectory in Physics

Instantaneous Velocity - Formula based Examples for JEE

Learn About Angle Of Deviation In Prism: JEE Main Physics 2025

Other Pages
NCERT Solutions for Class 12 Chemistry Chapter 1 Solutions

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Solutions Class 12 Notes: CBSE Chemistry Chapter 1

NCERT Solutions for Class 12 Chemistry Chapter 6 Haloalkanes and Haloarenes

NCERT Solutions for Class 12 Chemistry Chapter 2 Electrochemistry

Electrochemistry Class 12 Notes: CBSE Chemistry Chapter 2




